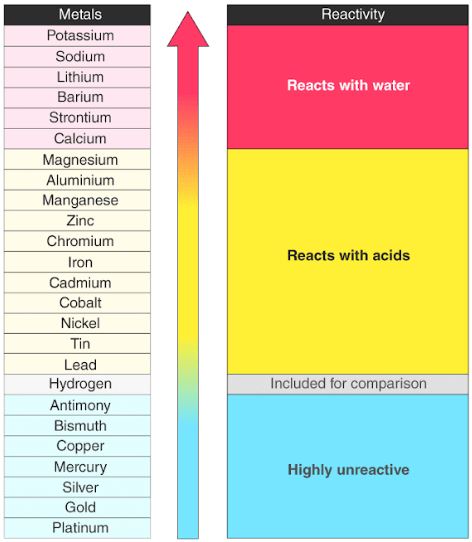
It is the electrons that are responsible for the chemical bavavior of atoms, and which identify the various chemical elements. In a neutral atom there are as many electrons as protons moving about nucleus. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The nucleus is composed of protons and neutrons. The atom consist of a small but massive nucleus surrounded by a cloud of rapidly moving electrons. K) 23.1 Thermal Conductivity 237 Specific Heat 0.9 Heat of Fusion 10.79 Heat of Vaporization 293.4 Atomic Number of AluminiumĪluminium is a chemical element with atomic number 13 which means there are 13 protons and 13 electrons in the atomic structure.The weaker electrostatic attraction of the electron being gained to the positive nucleus, makes it harder for the atom to gain an electron and it decreases in reactivity in group 7.Aluminium – Properties Element Aluminium Atomic Number 13 Symbol Al Element Category Poor Metal Phase at STP Solid Atomic Mass 26.9815 Density at STP 2.7 Electron Configuration 3s2 3p1 Possible Oxidation States -2 -1 +1 +2 +3 Electron Affinity 42.5 Electronegativity 1.61 1st Ionization Energy 5.9858 Year of Discovery 1825 Discoverer Oersted, Hans Christian Thermal properties Melting Point 660 Boiling Point 2467 Thermal Expansion µm/(m Therefore, the attraction of the outer electron to the nucleus decreases as you go down group 7. There are more electrons shielding the positively charged nucleus from the outer electron. They increase in electron shells so the atoms are larger as you go down the group. The atomic mass of the halogens increases.As you go down group 7, reactivity of the halogens decreases because: The negatively charged electron is attracted to the halogen atom due to the positive charge of the atom’s nucleus. 
Halogens react to gain an electron so they become more stable. Group 7: Reactivity (GCSE Chemistry) Group 7: Reactivity Halogens – trend in reactivityĪs you look at the trend in reactivity of group 7, the reactivity of halogens decreases so fluorine is the most reactive halogen and astatine is the least reactive halogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

/complete-periodic-table-of-elements-royalty-free-vector-166052665-5a565f0e47c2660037ab8aca.jpg)
 RSS Feed
RSS Feed
